Reverse Osmosis
Ideal for rural, commercial and industrial usage
What is Reverse Osmosis?
Reverse Osmosis is a technology used to remove dissolved solids or dissolved salts from water by pushing it under pressure through a semi-permeable membrane. The membrane as a filter is so fine that organic matter, colloidal solids or heavy metals, bacteria and viruses are also removed.
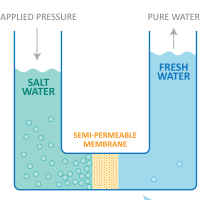
RO membranes can remove over 98% of dissolved salts/solids, over 99% of organic matter and colloidal matter and almost 100% of bacteria and viruses.
Ideal for rural, commercial and industrial usage
Specifications
| TECHNICAL SPECIFICATIONS | 6KLD | 12KLD | 18KLD | 24KLD | 30KLD+ |
|---|---|---|---|---|---|
| Water Production | 6,000 litres RO Water P/Day | 12,000 litres RO Water P/Day | 18,000 litres RO Water P/Day | 24,000 litres RO Water P/Day | 30,000 litres RO Water P/Day |
| Dimensions | 850W X 760D X 1670H | 2400W x 650D x 1585H | |||
| Membranes | Polyamide thin-film composite / Suitable Pressure Range 8 – 30 Bar | ||||
| 1 Membrane | 2 Membrane | 3 Membrane | 4 or 6 Membrane | 8 Membrane | |
|
Booster Pump + VFD Plus HMI |
Seamless SS Vertical 31 Stage 240V 3 Phase 3KW Pump - 50Hz / ABB VFD / Siemens HMI Seamless SS Vertical 24 Stage 240V 3 Phase 2.2KW Pump - 50Hz / ABB VFD / Siemens HMI |
||||
| Frame + Housing | Stainless Steel SUS304 Housing / Max Pressure Resistance 450 PSI | ||||
| Power Requirements |
240V 10A / 15A / 25A / DGPO 480V 2 Phase Rural Power Supply / 30A |
||||
| Operation Limits |
Max Feed Temp 35°C / Min Feed Temp 5°C / Ph 4 - 9 Max Feed Pressure 26 bar / Min Feed Pressure 2 bar |
||||
| Custom | Speak to our friendly team about our custom skid mounted or containerized RO plants capable of 10m3 - 50m3 p/hour | ||||
Process
Stage 1
Pre-Treatment
Pre-treatment of feed water to the Reverse Osmosis Unit is key to a successful water treatment plant and depending on the water supply quality, various pre-treatment systems are utilised ranging from filter media, sediment filters, and or chemical…The process is as follows:
Media Filtration
1
If required, removes high levels of suspended and colloidal solids
5 Micron Sediment Filter
2
Removes suspended and colloidal solids in the water such as sand, rust, clay & other sediments
Activated Carbon Filter
3
Removes free residual chlorine & organic molecules or dissolved matter
0.5 / 1 Micron Sediment Filter
4
Removes finer sediment and suspended solids as well as some bacteria
Ultraviolet (UV) Light
5
Neutralises bacteria and viruses with Ultraviolet rays
Stage 2
Reverse Osmosis
For the final polish, the pre-treated water is carried to the Reverse Osmosis Unit and pushed, under pressure, through the semi-permeable membrane. The membrane wall prevents any remaining dissolved salts and heavy metals in the water, such as Na, Cl, Mg, Ca, SO4, CO3, to name a few. This allows only pure water to pass through for collection.




